Regeneron Monoclonal Antibody Treatment
The treatment uses a cocktail of two monoclonal antibodies casirivimab and imdevimab known as Regen-Cov in the US that bind specifically to two different sites on the coronavirus spike. In combination the two Eli Lilly treatments seem to.

What Are Monoclonal Antibodies And Can They Treat Covid 19 Iav
It contains the monoclonal antibodies casirivimab and imdevimab and is the first injectable coronavirus antibody treatment approved by the FDA to prevent COVID.
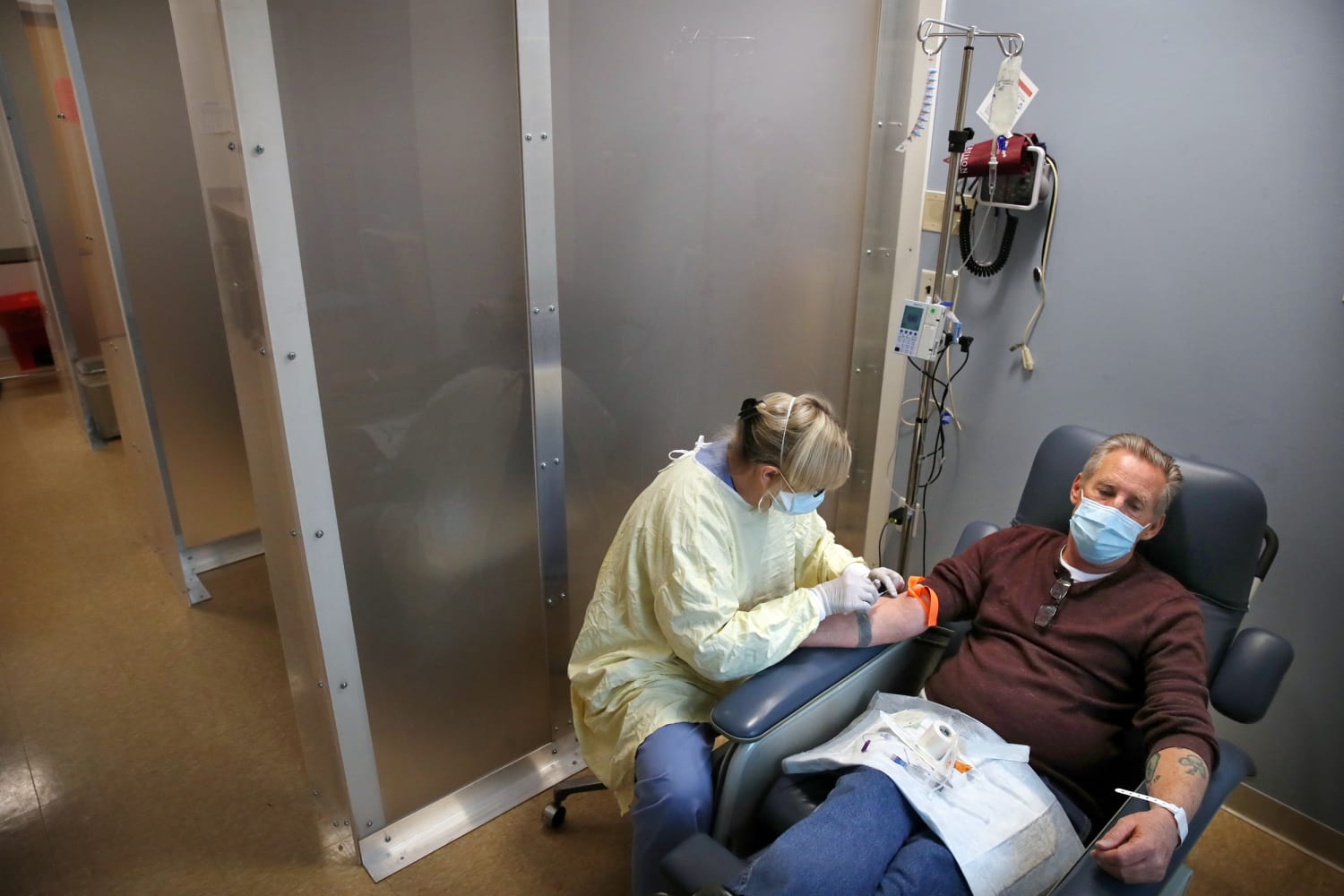
Regeneron monoclonal antibody treatment. I feel very lucky that the trial was up and running by the time I was taken to hospital with COVID-19 and I was able to receive. Casirivimabindevimab from Regeneron is a monoclonal antibody combination that is infused directly into a vein. The monoclonal antibody combination treatment from Regeneron has been authorized for emergency use by the FDA to treat mild to moderate COVID-19 in adults and pediatric patients 12 years of age and older weighing at least 40 kg 88 lbs with positive results of direct SARS-CoV-2 the virus that causes COVID-19 viral testing and who are at high risk for progressing to severe COVID-19 and.
The drug referred to as REGN-COV2 is the same one that President Trump received after being diagnosed with COVID-19 in early October. Read below to find out if monoclonal antibody treatment. What is monoclonal antibody treatment.
It is designed to block the spike protein in COVID-19 from attaching to and entering healthy. FDA authorizes REGEN-COV monoclonal antibody therapy for post-exposure prophylaxis prevention for COVID-19. There are currently two ways to receive this treatment.
Regeneron is also a major investment for one of DeSantis largest campaign contributors. Administration subcutaneous and the updated treatment indication including expanded high-risk criteria for Regeneron COVID-19 Monoclonal Antibodies. These antibodies are protective proteins that may help the body clear the virus faster and reduce symptoms when given soon after diagnosis.
Regeneron is responsible for development and distribution of the treatment in the US and Roche is primarily responsible for development and distribution outside the US. Health regulator authorized a lower dose of Regeneron Pharmaceuticals COVID-19 antibody cocktail that can be given by injection a. Prophylaxis with REGEN-COV is.
Multiple studies have confirmed that the Regeneron monoclonal antibody cocktail is effective against all major COVID-19 variants of concern including the Delta variant. Treatment This EUA is for the use of the unapproved product REGEN-COV casirivimab and imdevimab co-formulated product and REGEN-COV casirivimab and imdevimab supplied as individual vials to be administered together for the treatment of mild to moderate COVID-19 in adult and pediatric patients 12 years of age and older. On Friday November 21 the Food and Drug Administration FDA authorized the use of Regenerons monoclonal antibody cocktail for the treatment of COVID-19.
Regeneron is collaborating with Roche to increase global supply of REGEN-COV with expected production of at least 2 million treatment doses per year beginning in 2021. Monoclonal antibody treatments can be prescribed by health care providers to individuals 12 years of age and older who have been diagnosed with COVID-19 or who have been exposed to someone with COVID-19 and are at high risk for severe illness and hospitalization. Regeneron also called REGEN-COV is a mixture of two monoclonal antibodies.
Former President Donald Trump received Regenerons monoclonal antibody treatment when he. RECOVERY participant Kimberley Featherstone 37 was treated at Huddersfield Royal Infirmary and Calderdale Royal Hospital and randomly allocated to the monoclonal antiviral antibody combination. Bamlanivimab can still be used with etesevimab another monoclonal antibody treatment developed by Eli Lilly.
June 4 Reuters - The US. A monoclonal antibody treatment developed by the drug maker Regeneron sharply cut the risk of hospitalization and death when given to high-risk Covid-19 patients in a large clinical trial the. She said I was certainly glad to take part in the RECOVERY trial.

Monoclonal Antibody Treatment Idph
News Recovery Trial Monoclonal Antibody Treatment Saves Lives Of Seronegative Hospitalised Covid 19 Patients Nihr

Covid In Florida Gov Desantis Launches Monoclonal Antibody Treatment Centers Cbs Miami
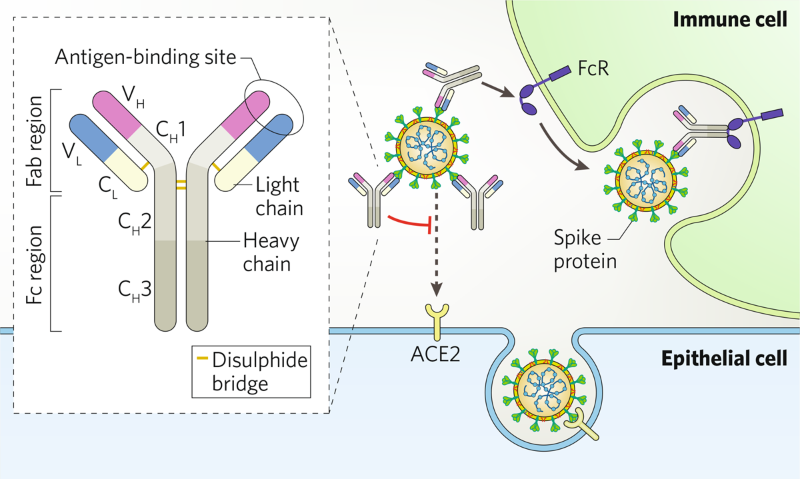
Hunting For Antibodies To Combat Covid 19

Regeneron S Covid 19 Antibody Cocktail Trial Releases Encouraging Early Data

Fda Authorizes Regeneron S Monoclonal Antibody Treatment For Covid 19 Rt A Medqor Brand

Experimental Covid Treatment Expands In Lehigh Valley The Morning Call
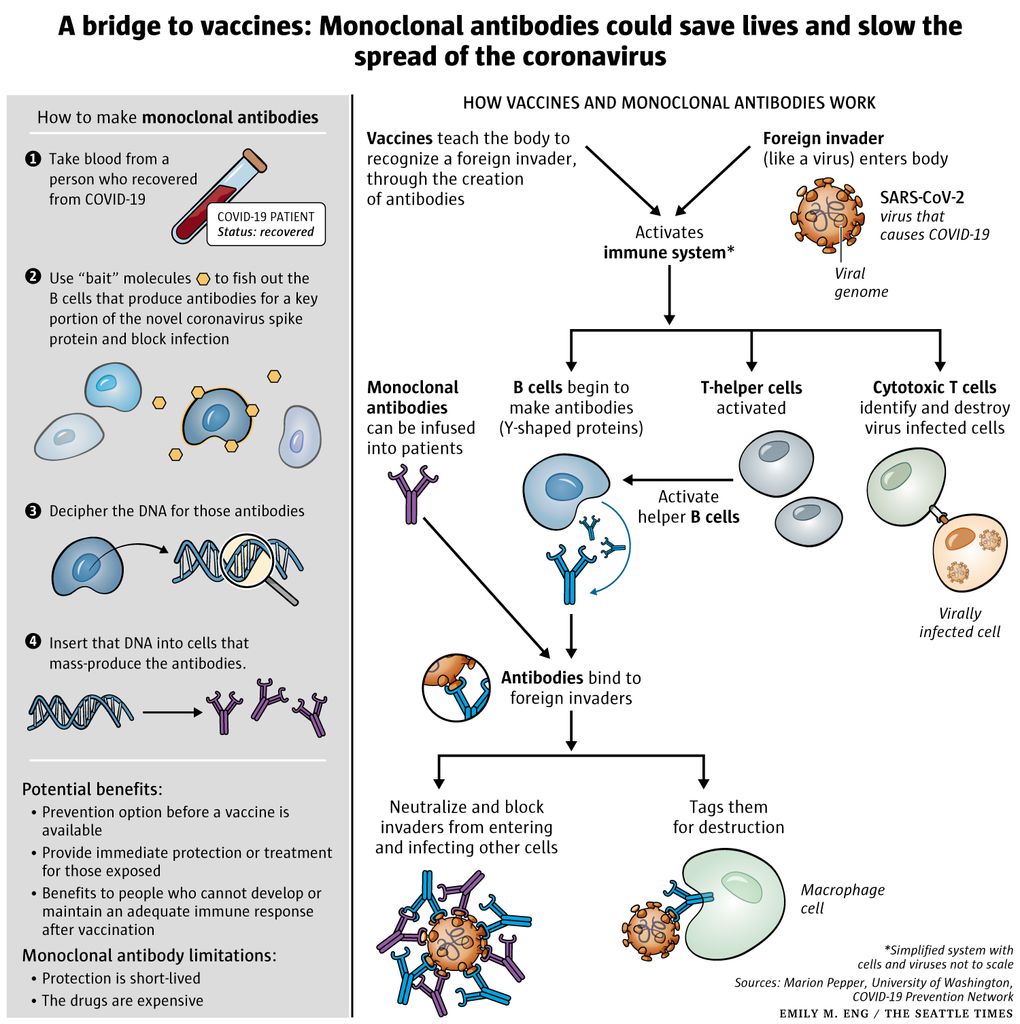
Monoclonal Antibodies Could Fill The Covid 19 Treatment Gap Until Vaccines Arrive But At A Cost The Seattle Times

Fda Approves Emergency Use Of Monoclonal Antibody For Treatment Of Covid 19
Devexplains Monoclonal Antibody Treatment For Covid 19 Devex

Monoclonal Antibodies Could Ease Record Covid Hospitalizations Why Are They Going Unused


:strip_exif(true):strip_icc(true):no_upscale(true):quality(65)/d1vhqlrjc8h82r.cloudfront.net/08-13-2021/t_bd75a5b6d5344ad6b4898feb6cdb4805_name_image.jpg)




/cloudfront-us-east-1.images.arcpublishing.com/gray/RUILPPSP6BDB5JTPJTAPSMFONA.jpg)
Post a Comment for "Regeneron Monoclonal Antibody Treatment"